BOM Sampling Plan
Sampling is a method widely used to define product meeting Quality Criteria.
Sampling is the selection of a representative portion of the inventory of an item to make inferences about the entire inventory. In pharmaceutical manufacturing, samples are drawn from different stages of the process for both controlling process parameters and assessing product quality.
Instead of waiting until production is in full swing, the sampling process allows you to catch discrepancies or failures to meet your specification standards when inventory is received. This can prevent a large number of wasted resources, including both time and money.
BOM Sampling PlanAQL can be set up on both Incoming Inspections as well as BOMs, but sampling plan settings do not cross over. Sampling plans set up on an Inspection Configuration will only apply to received materials, and plans set up on a BOM will only apply to production records. The setup on each page is fairly similar, see the BOM Sampling Plan page for exact steps.
Acceptable Quality Level (AQL)
DataNinja uses AQL "General Inspection Levels" as the parameter, and these values are already integrated into the system as pre-existing values. This means that DataNinja will do the math based on your:
- Lot size (quantity)
- Inspection levels/sampling plan (i.e. Reduced I, Normal II, or Tightened III).
- Defect classifications (i.e. Critical, Major, or Minor).
- AQL level corresponding to inspection & defect levels.
The user will be guided by the DataNinja to complete the inspection process after receiving inventory.
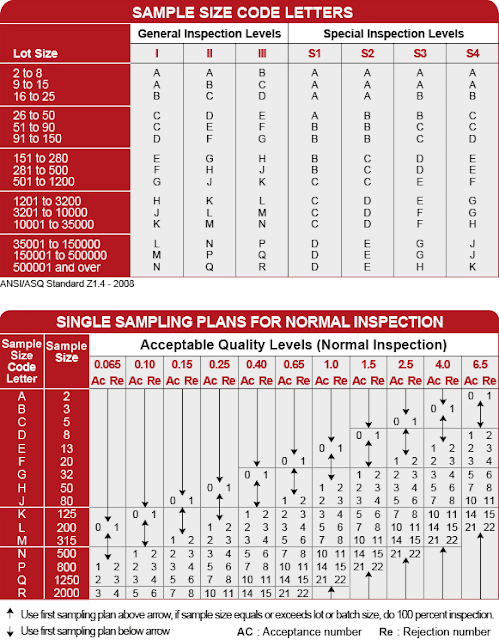
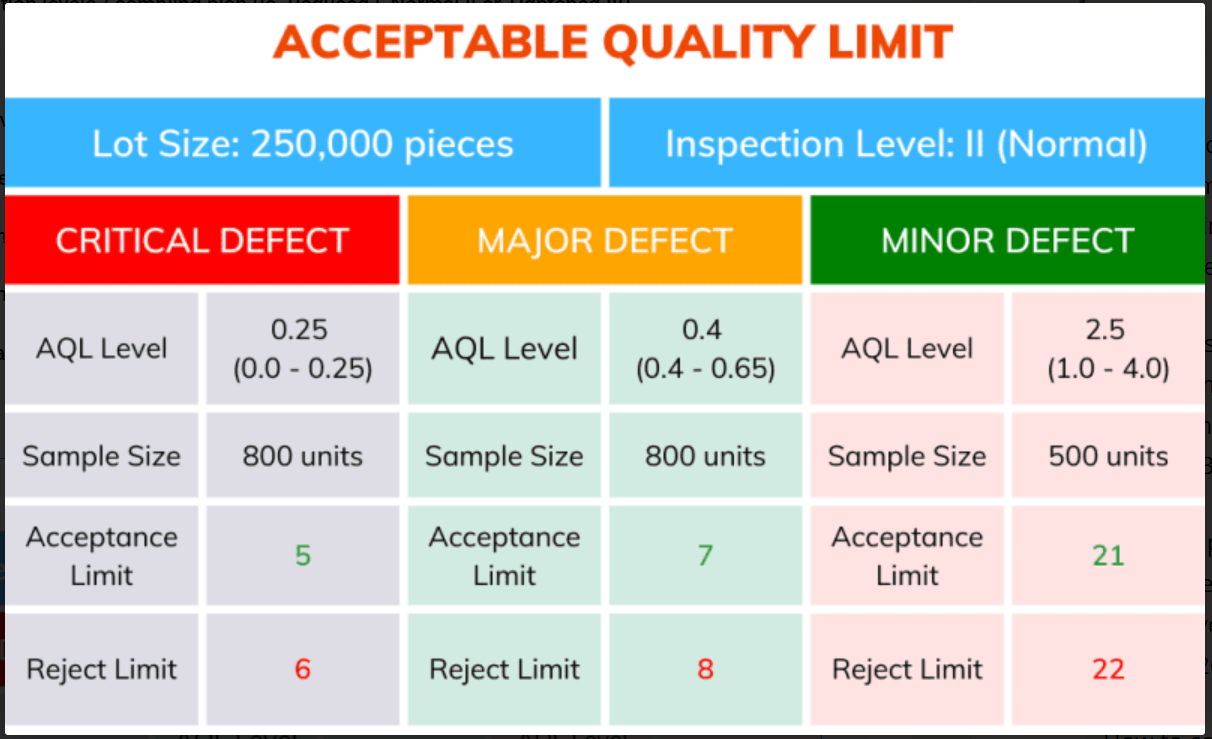
Based on the information above, a plan can be generated for sampling and acceptance limit. Below is an example for a lot of 250,000 on AQL II.
Setting Up AQL in DataNinja
Acceptable Quality Limit/Level (AQL) is the amount of deviation allowed in a product and is set on a per-part (or per-inspection configuration) basis. This built-in feature allows another level of quality control to make sure the product is at your exact specifications. DataNinja's Quality Sampling Plan feature will automatically calculate the amount of product that needs to be inspected, as well as how many of the inspected items can fail before a 100% inspection is required.
On a Bill of Materials
From the BOMs page, double click on the relevant BOM or click 'View' in the Actions column. Once the Edit BOM page opens, go to the Sampling Plan tab, click 'Add Sample Plan', and select which plan to use.
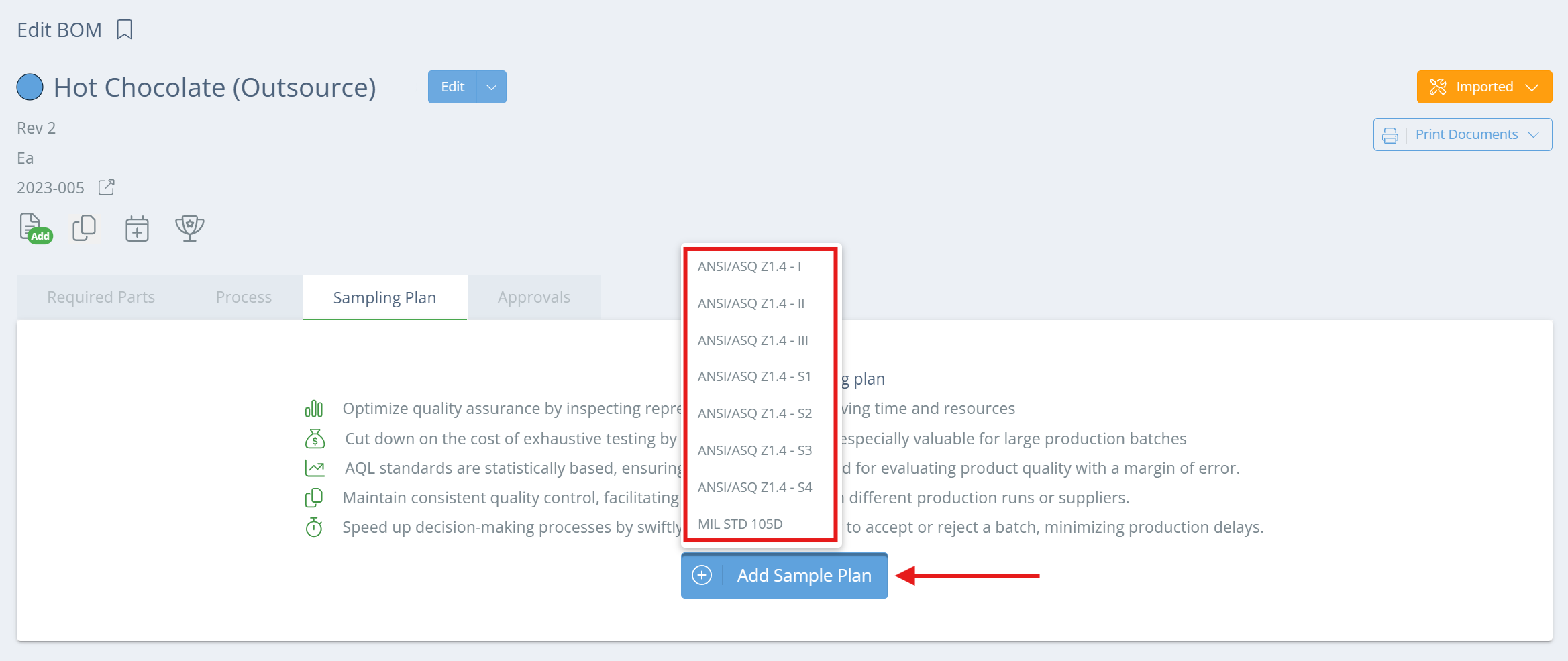
Screencap from v4.21.264.255
In the new aside, enter any instructions, and click 'Add Sampling Plan'. Then specify the AQL and any categories, and repeat as many time as needed. Multiple categories can be added to each Critical/Major/Minor sampling plan.
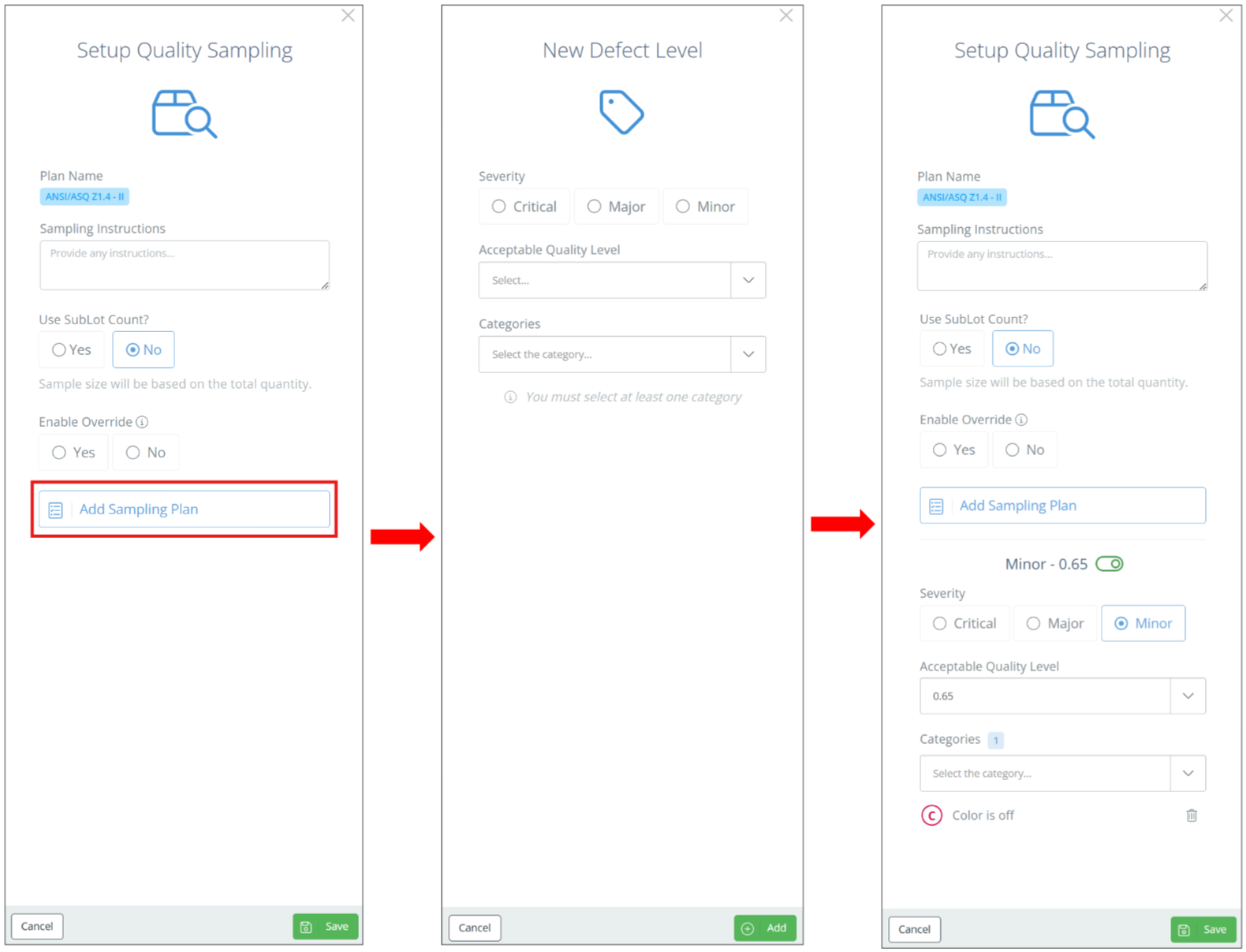
Screencap from v4.21.264.255
Once all desired severity levels have been added, click "Save" and the sampling plan will show on the Inspection Configuration page.
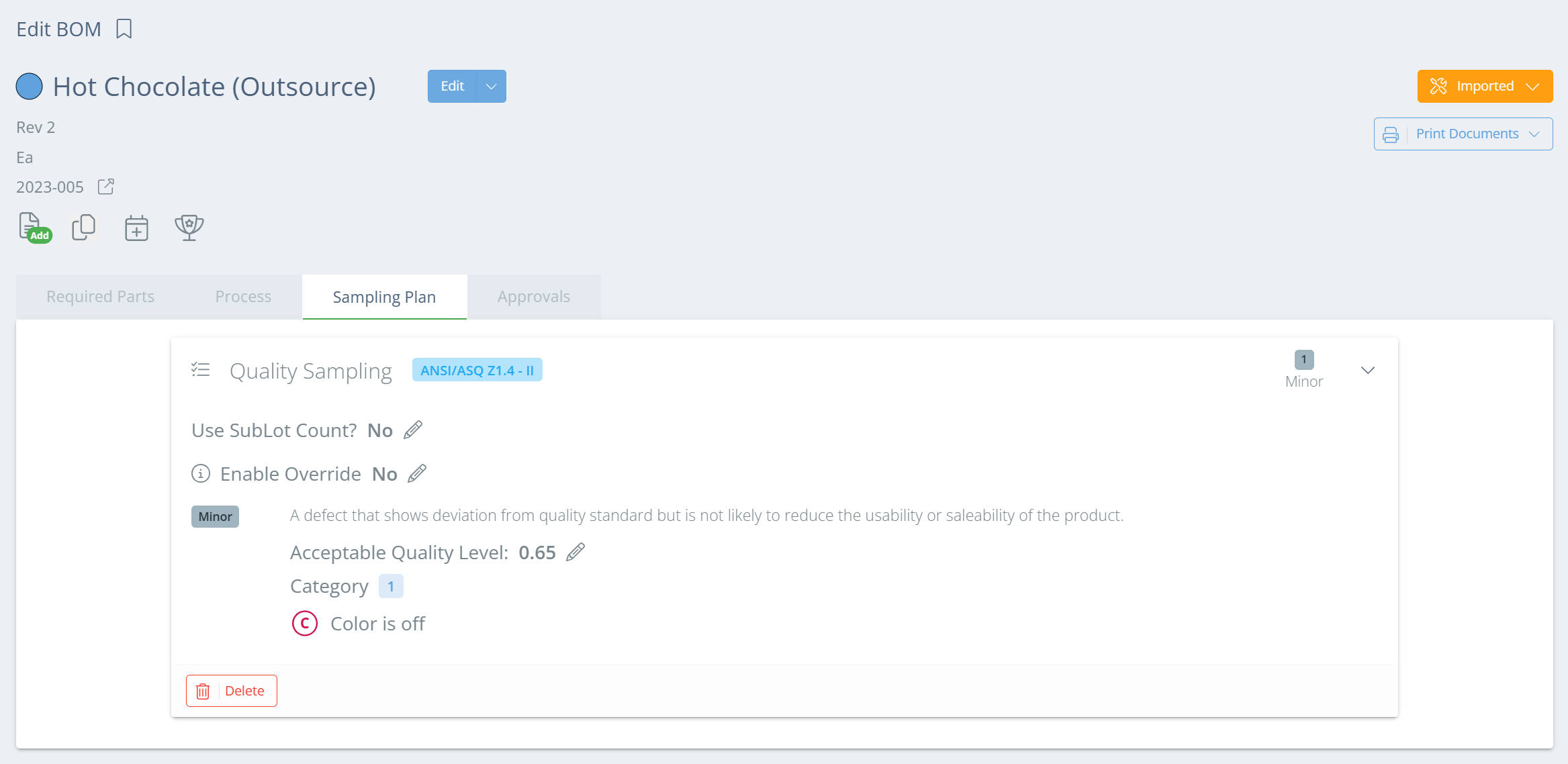
Screencap from v4.21.264.255
Sublot Count or Not?
If you select "Yes" to sublot count, the sample size will be based off the number of labels. If you select "No" then the sample size will be based on the total quantity produced in a batch instead. Choose whichever option best fits your needs and sampling requirements.
Enable Override (Optional)
If 'Yes' is selected, then users with the correct permissions can override the AQL requirements for this inspection/sampling.
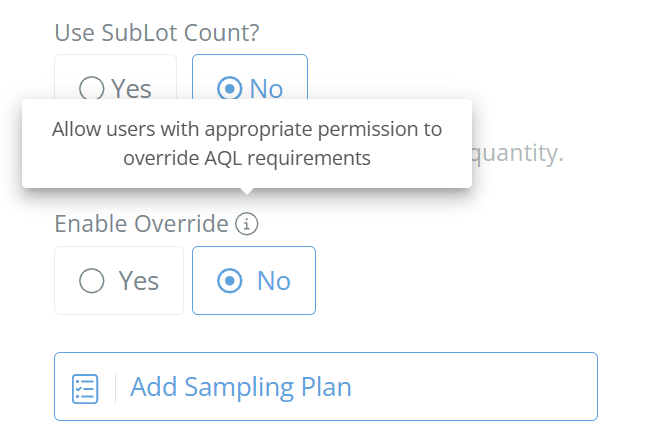
Screencap from v4.21.264.255
Updating a Sampling PlanYou can update a sampling plan until the BOM is released and scheduled. This action will lock the Manufacturing Master Record and can only be updated by creating a new version. Use our Copy a BOM page for directions.
Updated about 2 months ago
